|
SODIUM FLUOROSILICATE
| ||
|
PRODUCT IDENTIFICATION |
||
| CAS NO. | 16893-85-9 |
|
| EINECS NO. |
| |
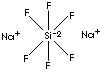
| FORMULA |
Na2SiF6 | |
| MOL WT. | 188.06 | |
|
H.S.CODE |
2826.20 | |
| TOXICITY | ||
| SYNONYMS | Sodium Silicofluoride; Sodium Hexafluorosilicate; | |
| Sodium Fluosilicate; Earwig Bait; Disodium Hexafluorosilicate; | ||
| RAW MATERIALS | fluorosilicic acid with sodium carbonate | |
|
CLASSIFICATION |
|
|
|
PHYSICAL AND CHEMICAL PROPERTIES |
||
| PHYSICAL STATE | white crystalline powder | |
| MELTING POINT | ||
| BOILING POINT | 500 C (Decomposes) | |
| SPECIFIC GRAVITY | 2.679 | |
| SOLUBILITY IN WATER | 0.76 g/100 ml at 25 C , 2.45 g/100 ml at 100 C | |
| pH | 3.0 - 4.5 (1% Sol.) | |
| VAPOR DENSITY | ||
| FLASH POINT | ||
|
APPLICATIONS |
||
|
Fluorine (Symbol : F; Atomic no. 9 ) is a yellowish, poisonous, corrosive gas under ordinary conditions. Fluorine becomes a yellow liquid upon cooling. It is the most reactive nonmetallic element and extremely powerful oxidizing agent. Because of its extreme reactivity, fluorine does not occur uncombined in nature. Fluorine occurs widely combined in the mineral fluorspar( fluorite, the chief commercial source), cryolite and apatite. The preparation of the free element is carried out by the electrolysis of a molten mixture of hydrogen fluoride, HF, and potassium fluoride, KF in the absence of water. Fluorine can be safely stored under pressure in cylinders of stainless steel if the valves of the cylinders are free from traces of organic matter. The outstanding oxidizing properties of the elemental gas are used in some rocket fuels. The element may be used for the fluorination of organic compounds with appropriate precautions. The element is used for manufacturing various fluorides including chlorine trifluoride ans cobalt(III) fluoride which are important fluorinating agents for organic compounds, sulfur(VI) fluoride used as a gaseous electrical insulator. Boron trifluoride and antimony trifluoride like hydrogen fluorides are important catalysts for alkylation reactions used to prepare organic compounds. Sodium fluoride (NaF) is used to treat dental caries and is often used for the fluoridation of drinking water to reduce tooth decay (However, there are reports of an accompanying risk of fluoride toxicity ). The element is also used for the preparation of uranium(VI) fluoride, utilized in the gaseous diffusion process of separating uranium-235 from uranium-238 (natural uranium) for reactor fuel. The importance of fluorine lies largely in its extreme ability to attract electrons and to the small size of its atoms, which can be attributed to form many stable complexes with positive ions like hexafluorosilicate(IV) and hexafluoroaluminate(III). Fluorine derivatives of hydrocarbons (compounds of carbon and hydrogen) are useful extensively as aerosol-spray propellants, refrigerants, solvents, cleansing agents for electrical and electronic components, and foaming agents in shipping-plastics manufacturing. Useful plastics with non-sticking qualities, such as polytetrafluoroethylene ( known by the trade name Teflon), are readily made from unsaturated fluorocarbons. A solution of hydrogen fluoride gas in water is called hydrofluoric acid, largely consumed for cleaning metals and for polishing, frosting, and etching glass. Hydrofluoric acid is also used as a catalyst for alkylation reactions. The chemical reactions are similar to those in the sulfuric acid process, but it is possible to avoid refrigeration. (In sulfuric acid alkylation, refrigeration is necessary because of the heat generated by the reaction). Sodium Fluorosilicate show similar application with sodium fluoride in the field of laundry souring, manufacturing enamels, manufacturing coated papers, wood preservative , foam production, opal glass, ore flotation and water fluoridation. |
||
| SALES SPECIFICATION | ||
|
APPEARANCE |
white crystalline powder | |
| ASSAY (Na2SiF6) |
98.0% min | |
| FLUORINE (F) |
59.0% min | |
| MOISTURE ( H2O) |
0.5% max | |
| WATER INSOLUBLES |
0.5% max | |
| HEAVY METAL ( as Pb) |
50ppm max | |
| SIEVE ANALYSIS | 98% min(40 mesh), 25% max (325 mesh) | |
| TRANSPORTATION | ||
| PACKING | 25kgs in Bag | |
| HAZARD CLASS | 6.1 | |
| UN NO. |
2674 | |
|
PRICES |
||